No content results match your keyword.
Content
You have successfully logged out.
Not registered yet?
Cyto-Set® Airstop
For Infusomat Space and Spaceplus Volumetric Pumps

Cyto-Set® AirStop is B. Braun’s closed system, specifically engineered to help prevent risk of exposure during administration of hazardous medicinal products (HMPs)1.
In combination with Chemfort® Closed System Transfer Devices2, this is the complete solution for the preparation and administration of HMPs, including Systemic Anticancer Therapy (SACT).
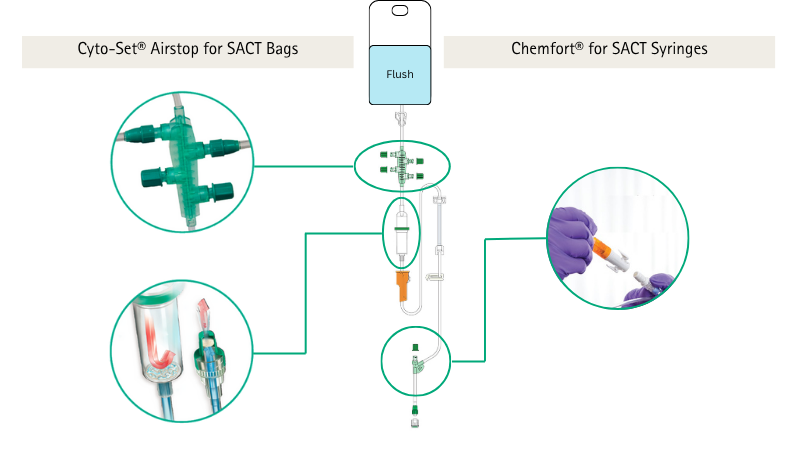
Components that emit an audible Click when securely connected, confirming the system is Closed and helping to Protect3 users from risk of exposure.
This is critical because there is no known safe level of exposure to HMP, therefore even one drop of HMP can be one too many4.

AirStop is a technology to help prevent air entering the line even if the drip chamber runs dry, for example during multiple infusion of Systemic Anticancer drugs. This helps to prevent therapy delays and improves workflow.
With components designed in the UK in collaboration with NHS hospitals, Cyto-Set® system is strictly aligned with the NHS Pharmaceutical Quality Assurance Committee’s Yellow Cover Guidance5, enabling administration of drug bags or bottles without the need for de-spiking or disconnection to help prevent the escape of hazardous medicinal products into clinical areas.

Cyto-Set® AirStop is a closed infusion system for Infusomat® Space and Spaceplus, designed to safely administer up to four bags or bottles of hazardous medicinal products.
A top spike and a tube with a clamp above the drip chamber allows giving set priming and flushing after each drug administration.
A green plate holds up to four needle-free connectors for secondary lines.
The drip chamber, with an AirStop filter, prevents air entering the line even if empty, therefore reducing therapy delays
Features include needle-free valves (e.g. for IV premedication) and a fill-stop cap (Prime Stop) at the patient connector.
The Proset Cyto-Set® line with Discofix allows the connection of hazardous medicinal product bags or bottles to the Cyto-Set® primary system.
Each bag or bottle requires its own secondary line. The Discofix stopcock prevents unintentional drug flow—like cytotoxic agents—before connection, reducing environmental contamination and protecting healthcare staff from exposure.

Cyto-Set AirStop technology helps prevent therapy interruptions—such as those caused by air entering the infusion line during multiple infusions—as well as reduce the risk of exposure to hazardous medicinal products and microbial contamination when addressing such issues.
Secondary Line with Discofix Stopcock: Enables dry connection to avoid accidental release of hazardous drugs (e.g. cytotoxic), reducing environmental contamination and protecting healthcare staff.
Needle-Free Valves: Built into the primary set to prevent chemical spills andminimise drug exposure risk.
Finger Stopper and Finger Grip on Needle-Free Valves
PrimeStop
Needle-Free Valve
Air Vent Filter and Back-Check Valve in the Spike
0.2 μm Sterifix® Filter:
Needle-free valve
1. Brünke, J., 2015. Evaluation of the chemical tightness of CSTDs. Hospital Pharmacy Europe, 79, pp.35-37. https://hospitalpharmacyeurope.com/clinical-zones/medicines-safety/evaluation-of-the-chemical-tightness-of-cstds/
2. P. Sessink et al., Validation of chemotherapy drug vapor containment of an air cleaning closed-system drug transfer device, Journal of Oncology Pharmacy Practice, 2022 Oct; 28(7):1508-1515
3. Marler-Hausen, T., Holt, C., Headley, C. and Sessink, P., 2020. Use of a closed-system drug transfer device reduces contamination with doxorubicin during bolus injection. British Journal of Nursing, 29(10), pp.S15-S21. https://www.magonlinelibrary.com/doi/full/10.12968/bjon.2020.29.10.S15)
4. Managing Hazardous Drug Exposure: Information for Healthcare Settings, Centers for Disease Control, page 3 last paragraph. https://www.cdc.gov/niosh/docs/2023-130/2023-130.pdf id=10.26616/NIOSHPUB2023130
5. Santillo, M., Field, A., Henderson, J., Hogan, A., Thoms, E. and Manomano, N., 2018. Guidance on handling of injectable cytotoxic drugs in clinical areas in NHS hospitals in the UK. NHS Pharmaceutical Quality Assurance Committee, London. https://www.sps.nhs.uk/wp-content/uploads/2018/07/Guidance-on-Handling-Cytotoxics-Ed-1-July-2018.pdf